Author(s): Peter F. Cook, Ashley Prichard, Mark Spivak, Gregory S. Berns
Published in Social Cognitive and Aective Neuroscience, Volume 11, Issue 12, (1 December 2016, Pages 1853–1862)
https://academic.oup.com/scan/article/11/12/1853/2544448
Dogs are hypersocial with humans, and their integration into human social ecology makes dogs a unique model for studying cross-species social bonding. However, the proximal neural mechanisms driving dog–human social interaction are unknown. We used functional magnetic resonance imaging in 15 awake dogs to probe the neural basis for their references for social interaction and food reward. In a first experiment, we used the ventral caudate as a measure of intrinsic reward value and compared activation to conditioned stimuli that predicted food, praise or nothing. Relative to the control stimulus, the caudate was signicantly more active to the reward-predicting stimuli and showed roughly equal or greater activation to praise vs food in 13 of 15 dogs. To confirm that these differences were driven by the intrinsic value of social praise, we performed a second imaging experiment in which the praise was withheld on a subset of trials. The dierence in caudate activation to the receipt of praise, relative to its withholding, was strongly correlated with the dierential activation to the conditioned stimuli in the first experiment. In a third experiment, we performed an out-of-scanner choice task in which the dog repeatedly selected food or owner in a Y-maze. The relative caudate activation to food- and praise-predicting stimuli in Experiment 1 was a strong predictor of each dog’s sequence of choices in the Y-maze. Analogous to similar neuroimaging studies of individual dierences in human social reward, our findings demonstrate a neural mechanism for preference in domestic dogs that is stable within, but variable between, individuals. Moreover, the individual differences in the caudate responses indicate the potentially higher value of social than food reward for some dogs and may help to explain the apparent efficacy of social interaction in dog training.
Introduction
As the first domesticated species, dogs have a unique relationship with humans. Dogs have been integrated into modern social life in many cultures, with millions serving as companion animals. As such, dogs benefit from a clear tendency of humans to bond socially with dogs (Odendaal and Meintjes, 2003; Beck and Madresh, 2008; Nagasawa et al., 2009; Stoeckel et al., 2014). But what is the nature of the relationship from the dog’s perspective? And, given the high degree of individual variability in dogs (see Scott and Fuller, 2012), how consistent across individuals are the biological underpinnings of this relationship? A better understanding of the proximal mechanisms driving dog–human interaction and the extent to which these vary across individuals will illuminate the dog–human social relationship. It is worth highlighting just how unique this cross-species relationship is. Although commensalism and symbiosis are not uncommon in the animal kingdom, a species-wide extension of social bonding mechanisms to include a wholly unrelated species is apparently very rare, and raises the intriguing possibility that human social behavior has served as a strong adaptive pressure in the evolution of domestic dog sociobiology (Reid, 2009). Quantifying the relative value of food vs. praise would also help inform ongoing and contentious debates regarding the most effective methods in dog training (e.g. McKinley and Young, 2003; Hiby et al., 2004; Blackwell et al., 2008).
Dogs are gifted at attending to, and interpreting, subtle human social cues (Lakatos et al., 2012; Merola et al., 2012; Müller et al., 2015), and a behavioral literature suggests that dogs act as if socially attached to humans (Topál et al., 1998; Palmer and Custance, 2008; although see: Prato-Previde et al., 2003; Rehn et al., 2013). Despite this, the motivations behind dog behavior toward humans can be difficult to disentangle from behavior alone. In terms of measuring preference, dog social behaviors are highly susceptible to prior patterns of food reinforcement (Bentosela et al., 2008; Elgier et al. 2009), and dogs frequently treat interaction with their owner as an avenue to acquire food (Cook et al., 2014a), even suppressing interest in food under communicative situations (Pongrácz et al., 2013). In direct tests of behavioral preference, some dogs select their owners and others food (Gácsi et al., 2005; Topál et al., 2005; Feuerbacher and Wynne, 2014, 2015)—but the behavior appears to be contingent on testing method, socialization history, reinforcement history and potentially many other factors including attention, stimulus salience and satiety. Further, although social reinforcement is a commonly used tool in dog training (Hiby et al., 2004), and many trainers believe it to be effective, it is quite difficult to experimentally isolate social and food reward in a training paradigm to measure their relative contribution to learning. Food delivery in dog training almost always includes a social component, and the acquisition rate of new behaviors can vary greatly depending on factors aside from reinforcement type. Notably, animals have long been known to show faculty for generalized learning (see Harlow, 1949), meaning that novel behaviors may be learned more quickly due to prior experience with even tangentially related learning tasks. The difficulties of isolating variables contributing to choice behavior and learning rate in purely behavioral paradigms highlight the potential value of a neurobiological approach seeking a consistent signal underlying individual differences in behavior.
Recent findings indicate that oxytocin, a neuropeptide critical for pair bonding within some species (Winslow, 1993; Young and Wang, 2004), has a role in mediating dog behavior toward humans (Odendaal and Meintjes, 2003; Romero et al., 2014; Nagasawa et al., 2015; Thielke and Udell, in press). Although oxytocin supports accounts of social attachment (although see Walum et al., 2016), the proximal neural mechanisms of social reward in the dog are unknown. In humans and other mammals, oxytocin receptors are dense in the ventral striatum, including the nucleus accumbens and caudate nucleus (Freund-Mercier et al., 1987; Olazabal and Young, 2006; Ross and Young, 2009), and these brain regions are also involved in social attachment in humans and other species (Rilling et al., 2002; Izuma et al., 2008; Young et al., 2008; Burkett et al., 2011). There is also strong evidence of a dissociation between the ventral and dorsal striatum, such that the ventral portion is more relevant to reward anticipation and learning (O’Doherty et al., 2004). In humans, activity in the ventral striatum has been associated with a wide variety of rewards, including both monetary and social (Delgado, 2007; Haber and Knutson, 2010; Lin et al., 2012; Pauli et al., 2016), and activation increases with increasing valuation (Knutson et al., 2000; Koeneke et al., 2008; Howe et al., 2013). Ventral striatal activity has also been shown to predict purchases (Knutson and Bossaerts, 2007), the choice of cola-drinks (McClure et al., 2004) and the popularity of songs (Berns et al., 2010). In animals, the ventral striatum has been shown to code the relative value of competing outcomes (Cromwell et al., 2005). Thus, there is good reason to believe that the analogous structure in dogs could be used to measure stable individual differences in relative preference for food and social rewards.
The advent of awake canine neuroimaging (Berns et al., 2012; Andics et al., 2014; for review see Cook et al., 2015) provides a unique opportunity to probe the neural mechanisms underlying the dog–human bond. Because of its coding of reward value, ventral caudate activation can serve as a measure of ‘choiceless utility’ (Loomes and Sugden, 1982), that is, the value a stimulus has for an individual who is not acting to acquire it. Choiceless utility tasks control for many of the confounds influencing active choice in dogs, and thus may be optimal for assessing the extent to which dogs value social interaction.
In dogs, we have previously shown a temperament-dependent increase in neural activity in the ventral caudate when dogs are presented with a stimulus associated with incipient receipt of food reward (Cook et al., 2015) and when they are presented directly with olfactory stimuli associated with familiar humans without linked reward (Berns et al., 2015). Although other brain regions may be differentially activated by reward preferences, no other region has the same strength of a priori justication for use with dogs (or any other animal) as does the ventral striatum (cf. Ariely and Berns, 2010 for Bayes’ factor of reward and striatum). If dogs are socially attached to humans and value interactions with them for more than the provision of food, social reward should be represented in ventral caudate activation in expectation of, and response to, human interaction. Moreover, the strength of this activation, relative to food, could be used as a marker for social preference. If so, then ventral caudate activation should be predictive of behavioral choice between food and social interaction.
In this study, we conducted three independent experiments. In Experiment 1, to identify a stable neural response associated with relative valuation of social interaction and food, we used functional magnetic resonance imaging (fMRI) to measure ventral striatal activation in 15 awake, unrestrained dogs while they passively viewed objects associated with incipient food reward or receipt of verbal praise from a primary handler. A neutral object, not previously associated with reward outcome, served as a control condition. Consistent with a choiceless utility approach, the primary experimental comparison was between activation during presentation of the food-predicting and praise-predicting conditioned stimuli, prior to delivery of reward.
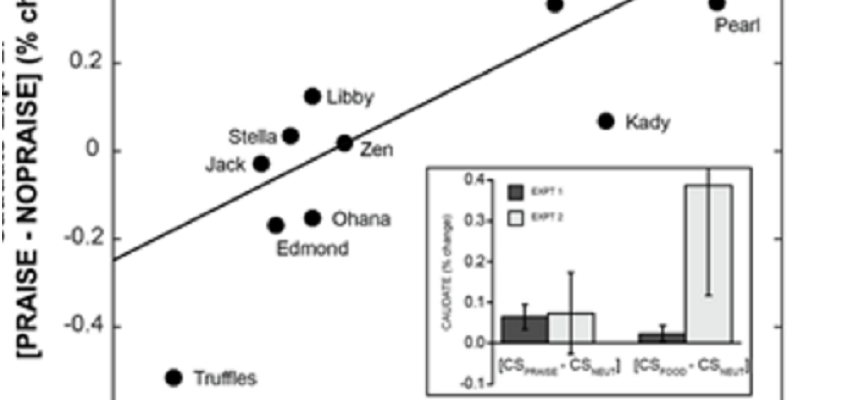
In Experiment 2, to determine stability of the neural valuation of praise within individual dogs, we replicated Experiment 1 with one alteration: on a subset of praise trials, the owner did not appear and praise the dog after the praise-predicting object was displayed. This withholding of praise constituted a violation of expectation, or a ‘negative prediction error’, as previously studied in humans (Schultz and Dickinson, 2000; McClure et al., 2003). Negative prediction errors result in a decrement of caudate activation, the magnitude of which is typically understood to relate to the value of whatever is being withheld. Experiment 2 allowed us to validate the relative neural response to praise and food from Experiment 1 using a complimentary experimental approach and a different temporal component of reward processing (expectation violation). Dogs who more greatly valued social reward ought to show greater positive activation to an object predicting incipient social reward (Experiment 1) and greater positive activation to receipt of praise vs. withholding of expected praise (Experiment 2).
In Experiment 3, to determine whether any stable neural marker of valuation could explain behavioral variability in active choice, each of the subjects took part in an independent, out-of-scanner behavioral choice task where they repeatedly selected between receipt of food or social interaction with their primary handler. Responses were modeled as relative stay and shift probabilities between food and handler, which could then be directly compared to relative caudate activations from Experiment 1.
Read the whole study and references here.
Want to read more from Aller Petfood’s News & Knowledge section?
Read the previous article here